Bayer Has Issued a Recall of Certain Nasal Sprays Due to a Potential Danger to Children
The recall impacts 786,100 nasal sprays.
Published May 4 2026, 3:25 p.m. ET

Cold and allergy sufferers know that sometimes, a high-quality nasal spray can help offer relief to those with congestion and stuffy noses. And while these types of medicines are typically sold over the counter (although there are prescription options available), they are regulated by the U.S. Food and Drug Administration to ensure that they meet the standards required.
Now, another agency is spreading the warning about a specific nasal spray, after it says a flaw was discovered.
This flaw has prompted the U.S. Consumer Product Safety Commission (CPSC) to share a recall of Bayer's nasal spray, after it says the child-resistant packaging failed to meet federal standards.
The recall includes a specific version of the nasal spray, which was sold to almost a million people in the U.S. Keep reading to learn more about the recalled item, including what the CPSC says you should do if you have some of these nasal sprays at home in your medicine cabinet.
Bayer issues a recall of a popular nasal spray.
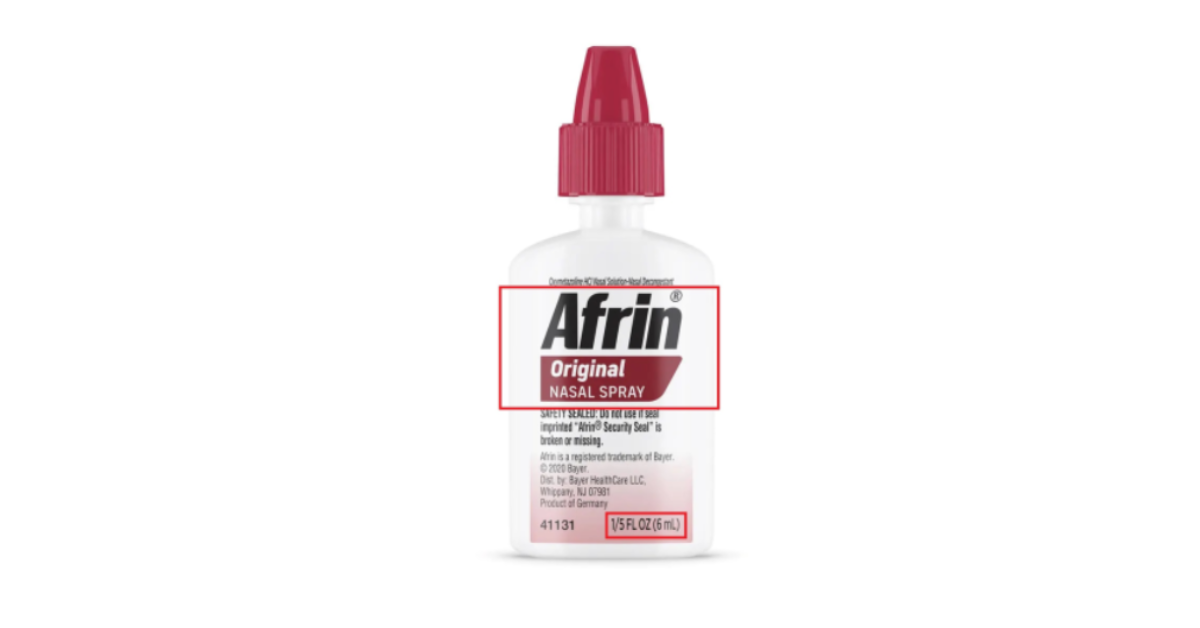
The CPSC issued a statement on Apr. 30, 2026, noting that Bayer had recalled a travel-sized version of its Afrin Original Nasal Spray Bottles, which were sold in 6 mL packages. The recall was initiated after federal regulators discovered that the label on the package and a lack of a proper child-resistant cap made the medication unsafe to be used in households with children under the age of six.
The concern is over a specific ingredient, known as imidazoline, which can be harmful if swallowed.
The recall includes the following lots:
- 230361
- 240822
- 241198
- 250066
- 250152
- 250646
- 250831
The bottles all include a label that says “Afrin® Original Nasal Spray” and “1/5 FL OZ (6 mL)," according to the CPSC statement. People were able to purchase these bottles nationally at airports and convenience stores from September 2024 to April 2026, according to the statement, which notes that they were sold for between $7 to $9 at the time they were on shelves.
What should you do if you purchased the recalled item?
Shoppers who have children in their home are being urged to reach out to Bayer through the company's website, which features an option to initiate a refund request. According to Bayer, the nasal spray should be photographed and then secured and put somewhere that children cannot reach them while you wait to receive a refund.
The statement also explains that there is nothing wrong with the formula itself, but instead the packaging, which makes it unsafe to be kept around children.
According to the Annals of Emergency Medicine, imidazolines can cause toxicity, which can only be fixed through medical intervention. When gone untreated, this type of toxicity can cause several complications, and in certain cases, even death.
If you have the recalled item in your home and it is somewhere that children can access it, you may want to secure it immediately, even if you don't currently have children living in your space with you at the moment.