Plastic Waste Gets a Second Life as Researchers Turn It Into Vinegar
Published March 5 2026, 8:55 a.m. ET

(L) A huge pile of microplastics; (R) University of Waterloo PhD student working on plastic upcycling method. (Cover Image Source: (L) Getty Images | Nugroho Ridho; (R) University of Waterloo)
Plastics are one of the most stubborn pollutants, slowly eating at the environment. Finding a way to recycle or destroy them is a dream of several conservationists. Now, like a knight in shining armor, this new study might bring them one step closer to that dream. Scientists have found a way to turn plastics into acetic acid using a source that's present in abundance: sunlight. Researchers at the University of Waterloo have evidently cracked the code to turning plastics into vinegar. This new approach is an efficient way of reducing plastic waste that's become a huge environmental problem of late. “Our goal was to solve the plastic pollution challenge by converting microplastic waste into high-value products using sunlight,” Dr. Yimin Wu, a professor of mechanical and mechatronics engineering and the Tang Family Chair in New Energy Materials and Sustainability, said in a statement.
The study, published in the journal Advanced Energy Materials, was led by Waterloo PhD student Wei Wei under the guidance of Wu. The university statement emphasized the increasing plastic waste disposal, especially microplastics, across the Earth's ecosystem. Microplastics are hardly noticeable, allowing accidental ingestion for both terrestrial and marine animals. In the aftermath, they can face toxic contamination, internal injuries, and much more that could even turn deadly. Even humans are heavily exposed to microplastics in everyday life, making them equally vulnerable to their harmful consequences. With hopes of finding a solution to this problem, the research team developed a bio-inspired cascade photocatalysis using iron atoms within carbon nitrate.

Waterloo PhD student Wei Wei, who led the research, works in the lab on plastic upcycling.
Once in contact with sunlight, a series of chemical reactions is triggered, which eventually leads to the conversion of plastic polymers into acetic acid or vinegar. The results were exceptional, increasing the hopes of breaking down the stubborn environmental plastics. One major aspect of the experiment was that it was conducted in water. Hence, the results provide a blueprint of how this technology would work in water bodies. The best part of this approach is that the conversion creates something edible instead of producing a gaseous byproduct. Since acetic acid is widely used in food production, chemical manufacturing, and energy applications, this approach, if successful on a large scale, would generate the least amount of waste.
According to the findings of this research, the popularly used acetic acid can be created from common plastic wastes. Plastics like polyvinyl chloride (PVC), polypropylene (PP), polyethylene (PE), and polyethylene terephthalate (PET) that are often found mixed in environmental waste can also be reduced to the aforementioned edible byproduct. Scientists believe that this approach to plastic waste management is more efficient than plastic incineration, which generates harmful gases. It holds great potential to be a widely used plastic upcycling method. “Both from a business and societal perspective, the financial and economic benefits associated with this innovation seem promising,” said Roy Brouwer, executive director of the Water Institute and a co-author of the article.
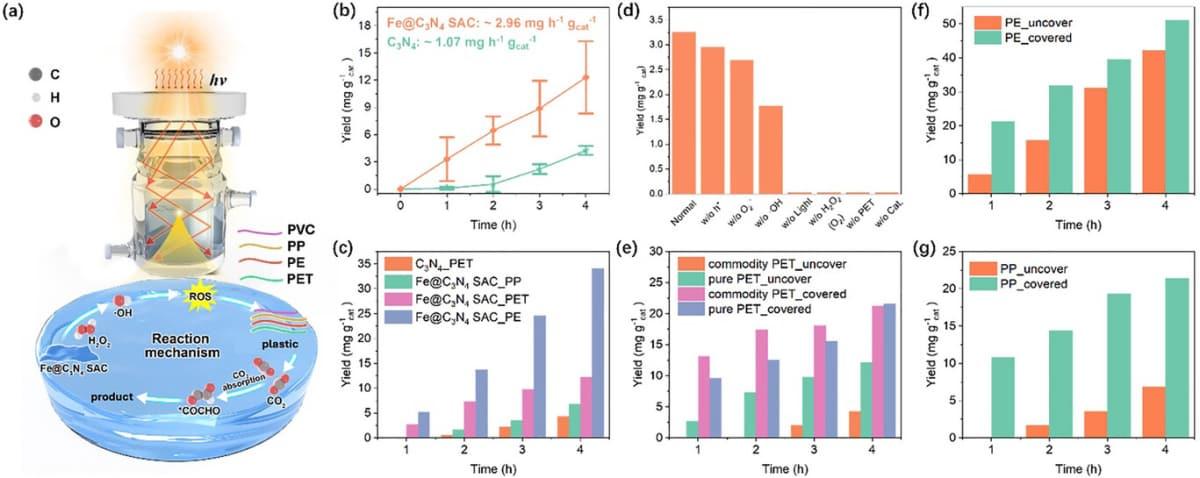
Demonstration of the plastic upcycling method.
“This method allows abundant and free solar energy to break down plastic pollution without adding extra carbon dioxide to the atmosphere,” added Wu. More importantly, this method breaks down microplastics at the chemical level, unlike the incineration process. Therefore, researchers have hope that the approach might help them remove or at least minimize microplastic accumulation in water systems. That being said, the method is still at a fetal stage, restricted to laboratory experiments. But with the determination of the researchers and the mission aligning with the university's Global Futures initiative, there's hope about the method proving groundbreaking in large-scale experiments as well.